 
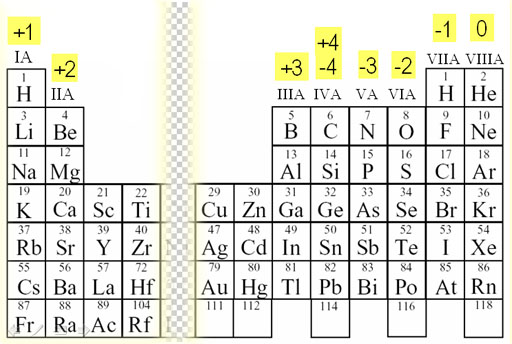
If the central atom is in its highest oxidation state, the names for these ions tend to end with the suffix '-ate'.įor example: CO 3 2- Carbon ate, SO 4 2- Sulph ate (containing sulphur(VI)), The overall charge on these ions is the sum of the oxidation number for the central atom and those for the oxygens present. 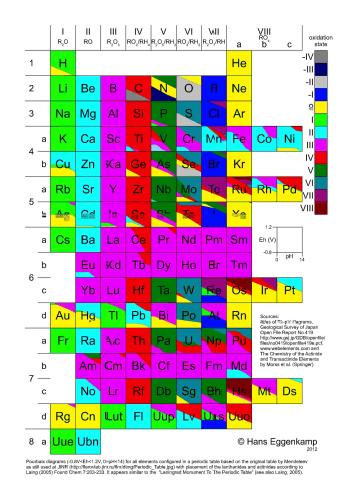
There are a range of molecular anions called 'oxo ions' which contain the atoms of an element bonded to several oxygen atoms. We can also have more complex cations (positively charged) and anions (negatively charged) which are molecules themselves, but carry an overall charge. In ionic compounds containing metal ions, the name is formed with the name of the metal (with the positive oxidation number), followed by the name of the non-metal (with the negative oxidation number) with the added suffix '-ide'.įor example: HCl - Hydrogen Chlor ide, NaCl - Sodium Chlor ide, Some of these prefixes are presented in the table below. If a given compound is multinuclear, that is, it consists of a single metal ion and a number of ions of another element then we use prefixes in front of the different parts of the name to denote how many of these atoms, ions or groups are present. In the complex 2-, the platinum has an oxidation state of +2 since the overall charge on the complex is -2 and there is a total ligand charge of -4, and -2 - (-4) = +2. Oxygen and sulphur as monatomic ligands have charges of -2, nitrogen -3, while ligands bearing no charge include water, ammonia (NH 3) and other amines and carbon monoxide (CO). For example, by definition, halide ligands, hydride, hydroxide (OH) and cyanide (CN) always have a charge of -1. In the chemistry of transition metal complexes, the oxidation number of the metal is defined as being the overall charge on the complex minus the charges of the ligand bonded to the metal. The lanthanides and actinides generally have +3 oxidation states though some +2, +4 and +6 states of some elements are known.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
